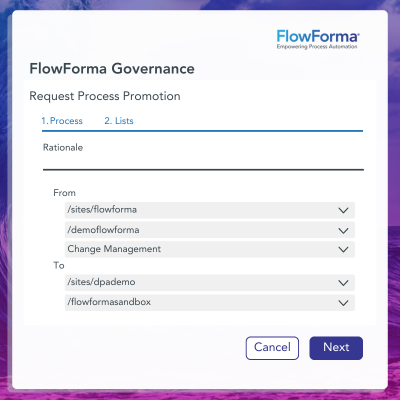
✔️ eSignatures: Enables users to construct an environment that can be independently audited against regulatory requirements.
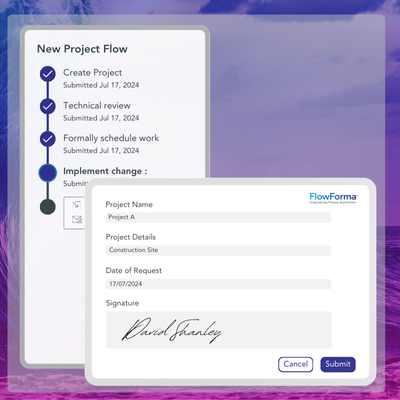
✔️ Integrated Form Audit Record: Processes can be designed so that process steps are signed, historical form activity is clearly visible, and a snapshot of the workflow is captured at a specific point in time.
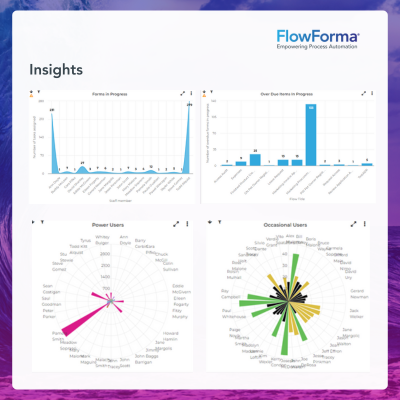
✔️ Flow Snapshots: All records can be easily and quickly understood by external auditors, who may not have any prior knowledge of processes in use.
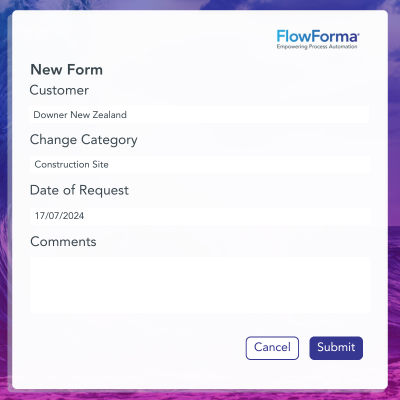
✔️ AI-powered Compliance: FlowForma's AI Copilot assists in building compliance workflows by automatically structuring processes using steps, questions, and rules.

.png?width=1000&height=226&name=G2%20Home%20Page%20Logos%20(2).png)














%20(1).png)